What Are Demodex Mites? Can Too Many Facial Mites Really Trigger Rosacea?
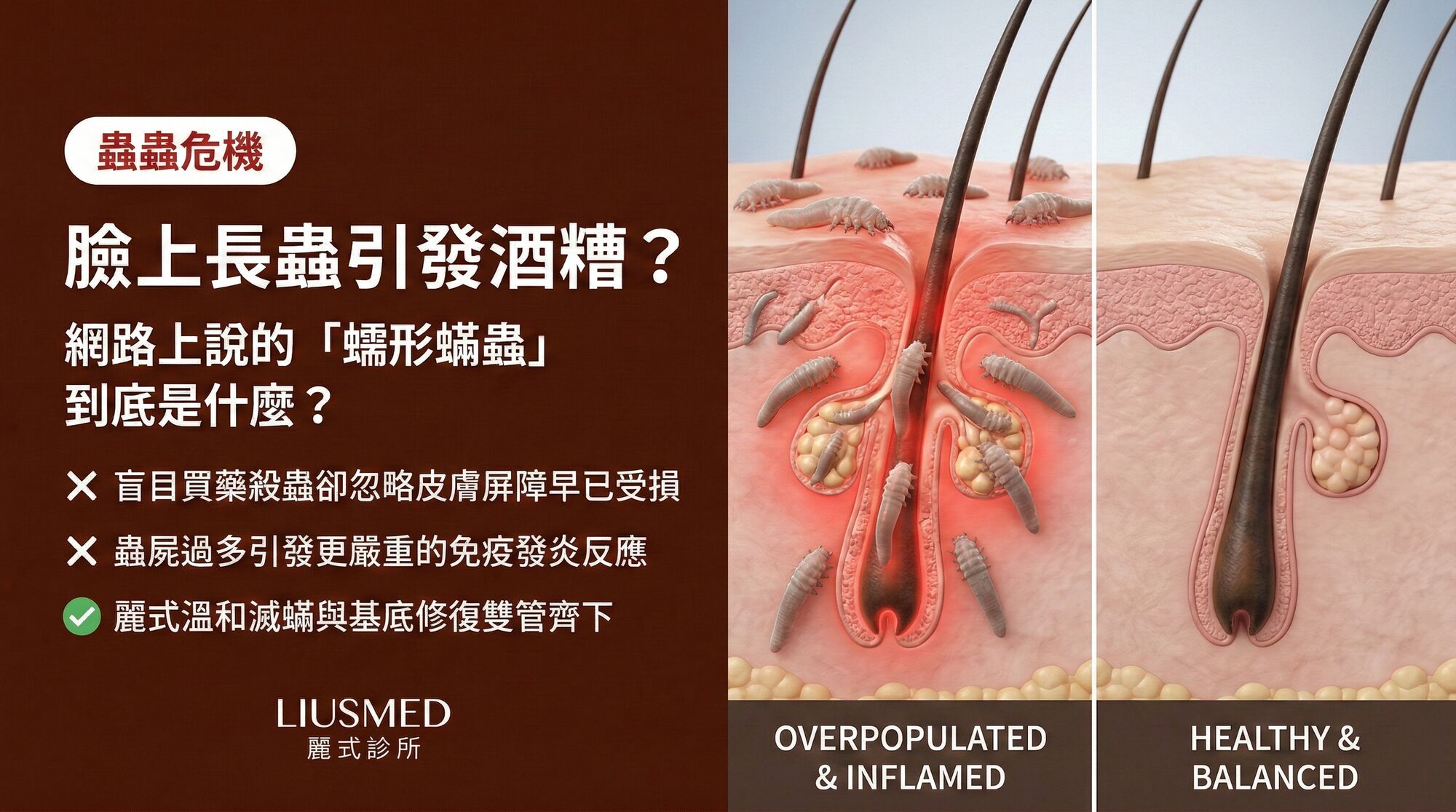
Right now, microscopic mites are living in your facial skin. This is normal. Demodex mites are commensal organisms found on virtually all adult human faces, typically causing no symptoms whatsoever. However, in rosacea patients, Demodex populations can expand to five to ten times the normal density, and at these elevated numbers, the mites overwhelm the skin's immune tolerance and trigger a sustained inflammatory response. The relationship between Demodex and rosacea has been debated for over a century, but modern immunological research has clarified the mechanism: it is not the mere presence of mites but their overpopulation, the bacteria they carry, and the immune responses their death provokes that drive rosacea pathology. This article explores the biology of Demodex, the critical density threshold, and how this knowledge translates into effective treatment.
Table of Contents
- Demodex Biology: Meet Your Facial Residents
- The Density Threshold: When Commensal Becomes Pathological
- Immune Activation: How Demodex Triggers Rosacea Inflammation
- The Bacillus Oleronius Connection
- Diagnosis: Detecting Demodex Overpopulation
- Treatment: Reducing Demodex to Restore Balance
- Frequently Asked Questions
- About the Author
- Disclaimer
Demodex Biology: Meet Your Facial Residents
Two species of Demodex mites colonize human facial skin: Demodex folliculorum and Demodex brevis.
Demodex folliculorum is the larger of the two (0.3-0.4 mm in length) and lives in the hair follicle infundibulum, the upper portion of the follicle near the skin surface. These mites feed on sebum and epithelial cells lining the follicle. They tend to cluster in groups, with multiple mites occupying a single follicle, particularly in areas with high sebaceous gland density: the nose, cheeks, forehead, and chin.
Demodex brevis is smaller (0.15-0.2 mm) and burrows deeper, inhabiting the sebaceous glands themselves. D. brevis tends to be solitary, with one mite per gland. Its deeper location makes it harder to detect on superficial sampling and potentially harder to treat.
Both species complete their entire life cycle on the human host over approximately 14 to 16 days. They are photophobic (avoid light) and are most active at night, when they emerge from follicles to mate on the skin surface before returning. They lack an anus and accumulate waste products internally throughout their lifespan, releasing them upon death. This detail is biologically significant, as we will see.
Demodex mites are acquired through direct facial contact, likely from parent to child during infancy and early childhood. By adulthood, colonization is nearly universal. Healthy adults typically harbor a density of 0.7 to 1.0 mites per square centimeter of facial skin, a level that the immune system tolerates without generating an inflammatory response.
The Density Threshold: When Commensal Becomes Pathological
The critical question in the Demodex-rosacea relationship is not whether mites are present (they almost always are) but whether their density has exceeded the threshold at which the immune system can no longer maintain tolerance.
Studies consistently show that rosacea patients have significantly higher Demodex densities than age-matched controls. The commonly cited clinical threshold for demodicosis is greater than 5 mites per square centimeter of skin, measured by standardized skin surface biopsy (SSSB). Some studies report mean densities of 10 to 20 mites per square centimeter in active papulopustular rosacea.
Why does density matter so much? Several factors converge:
Antigen load: Each mite presents its surface proteins, metabolic products, and eventually its entire internal contents to the host immune system. At low densities, the total antigen load is manageable and the immune system maintains tolerance. At high densities, the sheer volume of foreign antigen overwhelms tolerogenic mechanisms and tips the balance toward inflammation.
Bacterial load: As Demodex density increases, so does the population of associated bacteria (particularly Bacillus oleronius). The total bacterial antigen load scales with mite numbers, compounding the immune stimulus.
Physical follicular damage: High mite densities cause physical distension and disruption of follicular architecture. Multiple mites packed into a single follicle stretch the follicular wall, potentially causing micro-ruptures that expose dermal tissue to follicular contents, triggering foreign-body-type inflammatory responses.
Mite death and decomposition: With more mites, there are more mite deaths per unit time. Each dead mite releases accumulated waste products, partially digested host tissue, and bacterial contents into the surrounding skin. This bolus of inflammatory material is a potent activator of innate immune pathways.
Disruption of the skin barrier: High Demodex densities in the follicles compromise the follicular portion of the skin barrier, increasing transepidermal water loss and allowing environmental irritants to penetrate more easily. This barrier disruption synergizes with the direct inflammatory effects of the mites.
The density threshold concept explains why treatments that reduce Demodex numbers (rather than attempting complete eradication) can be clinically effective: the goal is to bring the population back below the tolerance threshold, restoring the commensal relationship.
Immune Activation: How Demodex Triggers Rosacea Inflammation
The immune response to Demodex overpopulation involves both innate and adaptive immune pathways, with the innate immune system playing the primary role in initiating and sustaining inflammation.
Toll-like receptor 2 (TLR2) activation: TLR2 is a pattern recognition receptor on keratinocytes, macrophages, and dendritic cells that recognizes microbial-associated molecular patterns (MAMPs). Demodex chitin (the structural polysaccharide of the mite exoskeleton), mite surface proteins, and bacterial components from the mite-associated microbiome all activate TLR2. This receptor is already known to be overexpressed in rosacea skin, creating a state of heightened sensitivity to these Demodex-derived signals.
KLK5-cathelicidin pathway amplification: TLR2 activation in rosacea skin triggers a cascade that is central to rosacea pathophysiology. TLR2 signaling upregulates kallikrein-5 (KLK5), a serine protease that cleaves the cathelicidin precursor protein (hCAP18) into the active peptide LL-37. LL-37 at normal levels is antimicrobial, but at the elevated concentrations produced in rosacea, it becomes pro-inflammatory: it activates formyl peptide receptors on immune cells, induces chemokine production, promotes angiogenesis, and directly activates TRPV1 (Transient Receptor Potential channels) channels on sensory nerves, contributing to neurogenic inflammation and flushing.
NLRP3 inflammasome activation: Demodex-derived chitin and the crystalline components of dead mites can activate the NLRP3 inflammasome, a cytoplasmic protein complex that processes pro-IL-1beta and pro-IL-18 into their active, secreted forms. IL-1beta is a potent pro-inflammatory cytokine that drives neutrophil recruitment, tissue remodeling, and fever-like local inflammation. Inflammasome activation helps explain the intense neutrophilic infiltrate seen in papulopustular rosacea lesions.
Adaptive immune involvement: While innate immunity initiates the response, sustained Demodex overpopulation can engage adaptive immunity. T-helper 1 (Th1) and T-helper 17 (Th17) lymphocyte populations are expanded in rosacea lesional skin. Th17 cells produce IL-17, which recruits neutrophils and promotes antimicrobial peptide production, further amplifying the inflammatory loop. The development of adaptive immune responses to Demodex antigens may contribute to the chronic and relapsing nature of rosacea.
Mast cell involvement: Mast cells, which are increased in number in rosacea skin, are activated both directly by Demodex-derived products and indirectly through neuropeptides (Substance P, CGRP) released during neurogenic inflammation. Mast cell degranulation releases histamine, tryptase, TNF-alpha, and VEGF (Vascular Endothelial Growth Factor — new blood vessel signal), contributing to vasodilation, vascular permeability, and angiogenesis.
The Bacillus Oleronius Connection
The discovery of Bacillus oleronius inside Demodex mites added a critical piece to the Demodex-rosacea puzzle. This gram-negative bacterium was isolated from the interior of Demodex mites taken from rosacea patients and has since been demonstrated to play a significant role in driving the inflammatory response.
Bacterial proteins as immune triggers: Bacillus oleronius produces multiple proteins that stimulate peripheral blood mononuclear cells (PBMCs) from rosacea patients to produce elevated levels of pro-inflammatory cytokines (IL-8, TNF-alpha) compared to PBMCs from healthy controls. This suggests that rosacea patients have a heightened immune reactivity to these bacterial antigens.
Neutrophil chemotaxis: Bacillus oleronius proteins are potent neutrophil chemoattractants, drawing neutrophils to the perifollicular dermis and contributing to the formation of papules and pustules. This helps explain the neutrophil-rich nature of papulopustular rosacea lesions.
Amplification upon mite death: When a Demodex mite dies, its body wall disintegrates and releases Bacillus oleronius and its products into the surrounding tissue. This sudden burst of bacterial antigen triggers an acute inflammatory flare. Because the mite life cycle is approximately two weeks, waves of mite death produce cyclical inflammatory flares, which may correlate with the waxing and waning pattern many rosacea patients experience.
Biofilm formation: There is emerging evidence that Bacillus oleronius may form biofilms within follicles, creating a protected reservoir of bacteria that is resistant to topical antimicrobials and continuously stimulates the local immune response.
The Bacillus oleronius connection explains why Demodex reduction (through anti-parasitic treatment) reduces rosacea inflammation even in cases where the mites themselves might not be the sole driver: killing the mites simultaneously eliminates their bacterial cargo.
Diagnosis: Detecting Demodex Overpopulation
Several methods are used to assess Demodex density in clinical and research settings.
Standardized Skin Surface Biopsy (SSSB): A drop of cyanoacrylate adhesive is applied to a microscope slide, pressed firmly against the skin for one minute, then peeled off. The adhered superficial follicular contents, including Demodex mites, are examined under light microscopy. A density greater than 5 mites per square centimeter is considered diagnostic of demodicosis. This is the most widely used quantitative method.
Dermoscopy: In-vivo dermoscopy can sometimes visualize Demodex tails protruding from follicular openings (appearing as whitish, fine, thread-like structures) and characteristic Demodex-associated follicular plugging. This is a non-invasive screening tool but is less quantitative than SSSB.
Confocal microscopy: Reflectance confocal microscopy allows in-vivo visualization of Demodex mites within follicles without the need for biopsy. The mites appear as bright, ovoid structures with characteristic dimensions. This is the highest-resolution non-invasive method but is not widely available.
Skin biopsy: Histological examination of a punch biopsy can reveal Demodex mites within follicles and associated perifollicular inflammation. This provides the most definitive assessment but is invasive and typically reserved for diagnostically challenging cases.
In clinical practice, many dermatologists diagnose Demodex-associated rosacea based on the clinical presentation (central facial papulopustular rosacea with or without background erythema) combined with a therapeutic trial of anti-Demodex treatment, rather than requiring formal density quantification.
Treatment: Reducing Demodex to Restore Balance
The therapeutic goal is to reduce Demodex density below the pathological threshold, thereby reducing the antigen and bacterial load driving the inflammatory cascade. Complete eradication is neither achievable nor necessary.
Topical ivermectin 1% cream: Currently the first-line treatment for Demodex-associated papulopustular rosacea. Ivermectin is an avermectin derivative that causes paralysis and death of Demodex mites by binding to glutamate-gated chloride channels. Beyond its anti-parasitic action, ivermectin has direct anti-inflammatory effects: it inhibits NF-kB signaling, reduces IL-1beta and TNF-alpha production, and modulates the cathelicidin pathway. Applied once daily for 12 to 16 weeks, it produces significant reductions in inflammatory lesion counts. A transient worsening (die-off reaction) may occur in the first one to two weeks as mite death releases inflammatory contents.
Topical metronidazole 0.75-1%: While primarily anti-inflammatory, metronidazole also has some anti-Demodex activity. It is often used as first-line or adjunctive therapy and is well-tolerated.
Oral ivermectin: In severe or refractory demodicosis, oral ivermectin (200 mcg/kg as a single dose, repeated at two-week intervals) can be used. Oral dosing achieves higher tissue concentrations than topical application, potentially reaching D. brevis in the deeper sebaceous glands.
Tea tree oil (diluted): Terpinen-4-ol, the active component of tea tree oil, has demonstrated in-vitro and in-vivo activity against Demodex mites. However, undiluted tea tree oil is highly irritating to rosacea skin. Formulations containing 4% terpinen-4-ol or specially diluted tea tree oil products may be used as adjunctive therapy under medical supervision, but tolerability must be carefully monitored.
Comprehensive rosacea management: Demodex reduction alone may not fully control rosacea if other pathogenic components (neurovascular dysfunction, chronic innate immune dysregulation, barrier deficiency) are also active. Optimal management often requires a combined approach that addresses multiple disease drivers simultaneously. Rosacea Injection Treatment protocols can complement anti-Demodex therapy by targeting the broader inflammatory and neurovascular components of the disease at the tissue level.
Maintenance therapy: Because Demodex cannot be eradicated from the environment and recolonization is inevitable, maintenance therapy (such as ongoing once or twice weekly topical ivermectin) is often necessary to prevent density from climbing back above the pathological threshold.
| Treatment | Target | Route | Duration | Key Consideration |
|---|---|---|---|---|
| Topical ivermectin 1% | Demodex + inflammation | Topical (once daily) | 12-16 weeks initial; then maintenance | First-line; well-tolerated; die-off reaction possible |
| Topical metronidazole | Inflammation + mild anti-Demodex | Topical (1-2x daily) | 8-12 weeks | Good tolerability; adjunctive role |
| Oral ivermectin | Demodex (systemic) | Oral (single dose, repeat) | 1-3 doses at 2-week intervals | Reserved for severe/refractory cases |
| Tea tree oil (4% terpinen-4-ol) | Demodex | Topical | Ongoing | Irritation risk; requires careful formulation |
| Targeted injection therapy | Broad inflammatory cascade | Injection | Variable | Addresses multiple disease drivers |
Frequently Asked Questions
Q1: Does everyone have Demodex mites on their face?
Yes. Demodex colonization is nearly universal in adults. Studies using sensitive detection methods find Demodex on over 95% of adult faces. The mites are acquired through close facial contact in early life and remain as permanent residents. Having Demodex mites is normal and, at low densities, they cause no symptoms.
Q2: If Demodex are normal, why do only some people develop rosacea from them?
The development of Demodex-associated rosacea requires two factors: mite overpopulation and a genetically or immunologically predisposed host. Not everyone who develops high Demodex densities will develop rosacea, and some rosacea patients have normal Demodex counts but still have active disease driven by other mechanisms. The interaction between host immune genetics (particularly TLR2 and cathelicidin pathway variants), environmental factors (UV exposure, barrier damage), and Demodex density determines whether disease develops.
Q3: Can I see Demodex mites with the naked eye?
No. At 0.1-0.4 mm in length, Demodex mites are below the resolution of the unaided human eye. They can be seen under low-power microscopy (40-100x magnification). The follicular "plugs" or rough texture sometimes visible on close inspection of rosacea skin may reflect high mite density but are not the mites themselves.
Q4: Can washing my face more frequently reduce Demodex numbers?
Standard cleansing does not significantly affect Demodex populations because the mites reside deep within follicles and sebaceous glands, beyond the reach of surface cleaning. Aggressive scrubbing can actually worsen rosacea by damaging the skin barrier and triggering neurogenic inflammation. Gentle cleansing is recommended to maintain barrier integrity, but it is not an effective Demodex-reduction strategy.
Q5: I have heard that Demodex are more active at night. Should I apply treatment before bed?
Yes. Demodex mites are nocturnal and emerge from follicles to mate on the skin surface during nighttime hours. Applying topical ivermectin in the evening increases the likelihood that active, surface-dwelling mites will contact the medication. Additionally, the mites are more vulnerable when they are outside the protective environment of the follicle.
Q6: Will killing all the Demodex mites cure my rosacea permanently?
Complete eradication of Demodex is not achievable because recolonization occurs through environmental contact. Even if near-complete elimination were possible, rosacea is a multifactorial disease, and Demodex overpopulation is only one contributing factor. Other components, including neurovascular dysfunction, innate immune dysregulation, and barrier deficiency, would still need to be addressed. The goal is sustained reduction of Demodex density below the pathological threshold combined with management of the other disease drivers.
About the Author
Dr. Ta-Ju Liu is the founder of Liusmed Clinic, specializing in regenerative medicine and minimal incision surgery. With extensive experience in treating chronic inflammatory skin conditions, Dr. Liu takes a mechanistic approach to rosacea management that addresses all contributing factors, from Demodex overpopulation and immune dysregulation to neurovascular dysfunction and barrier repair. His clinical philosophy prioritizes understanding disease mechanisms and translating that understanding into targeted, effective treatments.
Disclaimer
This article is provided for educational and informational purposes only and does not constitute medical advice, diagnosis, or treatment. The information about Demodex biology and treatment reflects current medical understanding, but individual patient presentations and treatment responses vary. Consult a qualified healthcare provider for personalized evaluation and treatment recommendations. Do not begin, discontinue, or modify any treatment based solely on the information presented in this article.
Specialties
Credentials
- Kaohsiung Medical University, School of Medicine
- Attending Physician, Dermatology, Kaohsiung Chang Gung Memorial Hospital
- Attending Physician, Aesthetic Center, Kaohsiung Chang Gung Memorial Hospital
- Visiting Physician, Dermatology, Xiamen Chang Gung Hospital
- Visiting Physician, Aesthetic Center, Xiamen Chang Gung Hospital
"For every surgery, I strive to achieve the best outcome through the smallest incision and finest technique. Minimally invasive surgery is not just a technique — it's a commitment of respect to every patient."
Recovery from filler complications needs peer support too
Want to learn more?
Schedule a consultation for professional evaluation and advice

