The Advantage of Manual Tranexamic Acid Injection: Directly Blocking Angiogenesis and Inflammation in the Dermis
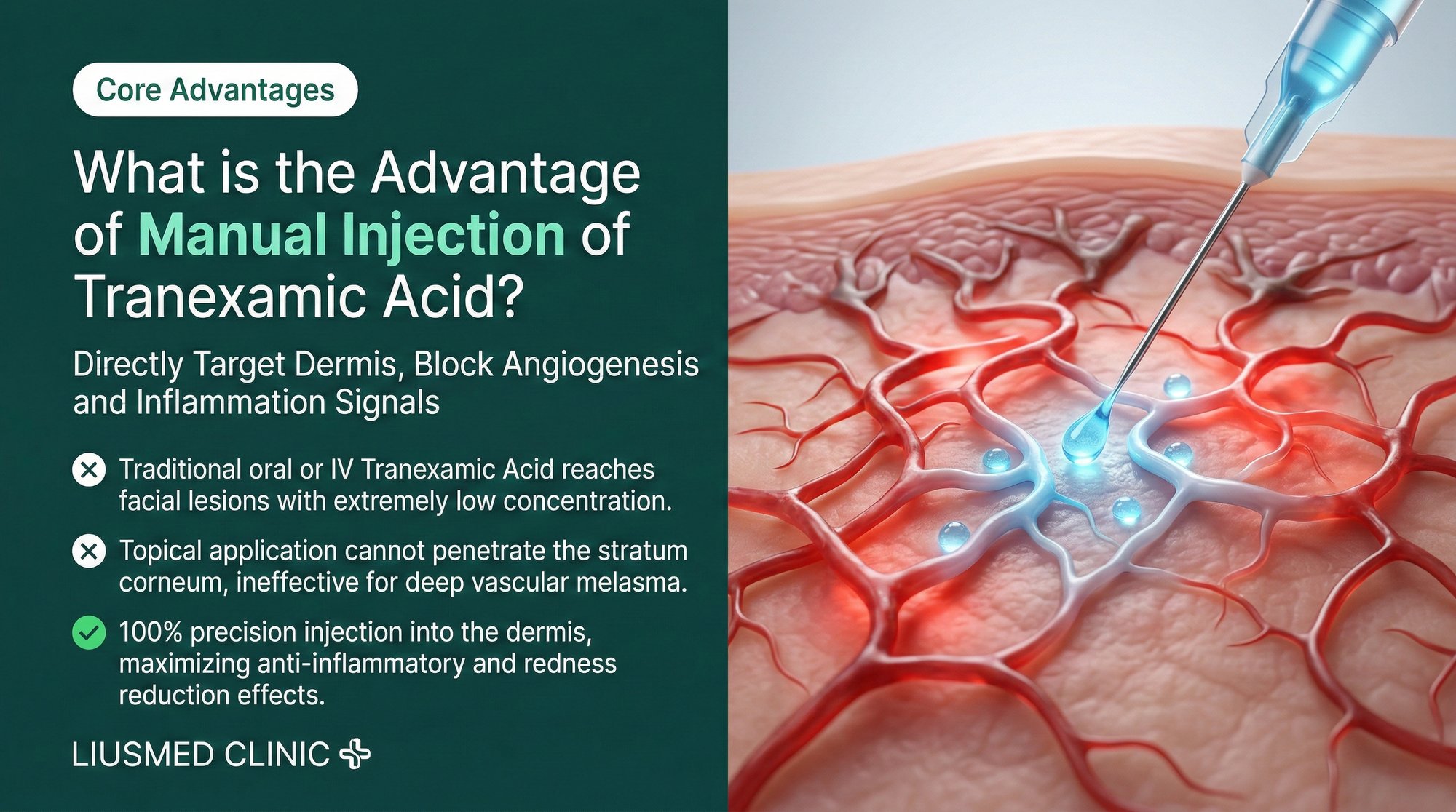
Tranexamic acid has quietly become one of the most effective weapons against melasma. But how it is delivered changes everything. Oral pills scatter a diluted dose across the entire body. Manual intradermal injection concentrates the full therapeutic payload exactly where the disease lives — in the inflamed, hypervascular dermis beneath the pigmented patch.
Table of Contents
- Tranexamic Acid: From Operating Room to Melasma Clinic
- The Vascular Dimension of Melasma: Why TXA (Tranexamic Acid) Works
- Oral vs. Topical vs. Intradermal: Three Delivery Routes Compared
- Manual Injection Technique: Precision at the Vascular Target
- Anti-Angiogenic and Anti-Inflammatory Mechanisms of Intradermal TXA
- Safety Profile and Practical Considerations
Tranexamic Acid: From Operating Room to Melasma Clinic
Tranexamic acid (TXA) was originally developed as a hemostatic agent — a drug that stops bleeding by inhibiting the breakdown of fibrin clots. For decades, its primary use was in surgery, trauma care, and the management of heavy menstrual bleeding. The molecule works by blocking the lysine-binding sites on plasminogen, preventing its conversion to plasmin.
The connection to melasma was discovered somewhat serendipitously. Clinicians noticed that patients taking oral TXA for menstrual bleeding experienced unexpected lightening of their melasma. This observation led to formal clinical investigation, and by the early 2010s, multiple studies had confirmed that oral TXA at low doses (typically 250 mg twice daily) produced significant improvement in melasma severity scores.
The mechanism linking a hemostatic drug to pigment reduction was not immediately obvious. But as researchers investigated further, they uncovered a crucial connection: the plasminogen/plasmin system is not only involved in clot breakdown — it also plays a central role in inflammation, angiogenesis, and melanocyte signaling. By inhibiting plasminogen activation, TXA simultaneously interrupts multiple pathological pathways that drive melasma.
This discovery reframed melasma as a condition with a significant vascular and inflammatory component, not merely a disorder of melanin production. And it raised an important question: if TXA works by acting on dermal blood vessels and inflammatory cells, why deliver it systemically when it could be placed directly at the target?
The Vascular Dimension of Melasma: Why TXA Works
The recognition that melasma has a vascular component was a paradigm shift in dermatology. Histological studies comparing melasma skin to adjacent normal skin have documented several consistent vascular abnormalities.
Melasma lesions contain a significantly higher density of blood vessels compared to non-lesional skin. These vessels are often immature, with increased permeability and a tendency to leak plasma proteins and inflammatory mediators into the surrounding dermis. Dermoscopic examination reveals characteristic arborizing telangiectasias within melasma patches that are absent in normal surrounding skin.
The molecular driver of this abnormal vascularity is VEGF (Vascular Endothelial Growth Factor — new blood vessel signal), which is found at elevated levels in melasma skin. VEGF is produced by keratinocytes, mast cells, and fibroblasts in response to UV radiation and inflammatory stimuli. It promotes the growth of new blood vessels (angiogenesis) and increases the permeability of existing vessels.
These leaky, proliferating vessels create a self-reinforcing cycle. They deliver more inflammatory cells and mediators to the dermis, which stimulates more VEGF production, which drives more vessel growth. The resulting inflammatory microenvironment continuously activates melanocytes, sustaining pigment overproduction regardless of surface-level treatments.
TXA interrupts this cycle at a critical junction. By blocking plasminogen activation, it reduces the conversion of pro-enzymes that activate VEGF signaling. It also directly inhibits the release of arachidonic acid and prostaglandins from keratinocytes, further dampening the inflammatory cascade.
Oral vs. Topical vs. Intradermal: Three Delivery Routes Compared
The effectiveness of TXA for melasma is well-established. The debate in clinical practice centers on the optimal delivery route. Each method has distinct pharmacokinetic properties that directly impact efficacy and safety.
Oral TXA is the most studied route. Standard dosing of 250 mg twice daily achieves systemic plasma levels that are sufficient to produce measurable melasma improvement. However, after oral absorption, hepatic first-pass metabolism, and distribution throughout the entire body, the concentration of TXA that actually reaches the dermal vasculature of a melasma lesion is a fraction of the administered dose. Furthermore, systemic use carries theoretical risks including thromboembolic events, though these are rare at dermatological doses.
Topical TXA avoids systemic exposure entirely. However, TXA is a hydrophilic molecule with poor percutaneous absorption. Studies measuring skin penetration of topical TXA formulations show that very little reaches the dermis, with the majority remaining on the skin surface or within the stratum corneum. Various delivery enhancement strategies (liposomal encapsulation, microneedling-assisted delivery) have been explored to improve penetration with variable success.
Intradermal TXA injection bypasses both limitations. By placing TXA directly into the dermis via manual mesotherapy, the full therapeutic dose reaches the target tissue. Local concentrations at the pathological site are orders of magnitude higher than what oral or topical delivery can achieve, while systemic exposure is minimal because only a small total dose is administered.
| Parameter | Oral TXA | Topical TXA | Intradermal TXA (Manual Injection) |
|---|---|---|---|
| Local dermal concentration | Low (systemic dilution) | Very low (poor penetration) | High (direct placement) |
| Systemic exposure | Moderate | Minimal | Minimal |
| Precision of delivery | None (whole-body distribution) | Surface only | Exact target depth and location |
| Onset of local effect | Delayed (absorption required) | Delayed and limited | Immediate |
| Risk of systemic side effects | Present (thromboembolic risk) | Negligible | Negligible |
| Frequency required | Daily oral dosing | Twice daily application | Every 3-4 weeks |
| Compliance burden | High (daily pills) | High (daily application) | Low (clinic visits only) |
Manual Injection Technique: Precision at the Vascular Target
The manual injection technique used in the Melasma Injection Treatment is specifically designed to deliver TXA to the superficial dermis where pathological angiogenesis and inflammation are concentrated.
The physician uses a fine-gauge needle (typically 30G or 32G) and performs serial puncture or nappage technique — a series of rapid, shallow injections spaced approximately 2 to 3 millimeters apart across the treatment area. The injection depth is controlled by the angle of needle insertion and the tactile feedback felt through the syringe.
This is where manual technique provides a decisive advantage over automated delivery systems. The superficial dermis is only approximately 0.3 to 0.5 mm below the skin surface. The margin between effective delivery and either too-superficial (epidermal bleb) or too-deep (subcutaneous) placement is measured in fractions of a millimeter.
An experienced practitioner can feel the tissue resistance change as the needle tip enters the papillary dermis. This real-time feedback allows continuous adjustment of depth, which is essential because skin thickness varies across facial regions: the periorbital skin may be less than 1 mm thick, while the malar cheek is significantly thicker.
The manual approach also allows the physician to modulate the injection volume and pressure based on the tissue response. Areas of dense fibrosis (common in long-standing melasma) require more volume and pressure to achieve adequate tissue diffusion, while thinner, more elastic areas require less. This adaptive delivery is impossible with standardized machine protocols.
Each treatment session delivers TXA directly to the perivascular space of the superficial dermal plexus — the exact location where pathological angiogenesis and vascular leak are occurring. This anatomical precision is the fundamental advantage of intradermal manual injection.
Anti-Angiogenic and Anti-Inflammatory Mechanisms of Intradermal TXA
When TXA reaches the dermal vasculature at therapeutic concentrations, it engages multiple interconnected mechanisms:
Plasminogen activation inhibition. TXA binds to the lysine-binding sites on plasminogen, preventing its conversion to plasmin by tissue plasminogen activator (tPA) and urokinase plasminogen activator (uPA). Plasmin is a potent activator of multiple downstream pathways including MMP (Matrix Metalloproteinase) activation, VEGF release, and pro-inflammatory cytokine processing. By blocking plasmin generation, TXA simultaneously reduces all of these pathological signals.
VEGF signaling reduction. With reduced plasmin activity, the proteolytic release of VEGF from extracellular matrix stores is diminished. Additionally, plasmin normally activates matrix metalloproteinases (MMPs) that further release matrix-bound VEGF. This dual blockade results in a significant reduction in local VEGF availability, slowing the formation of new pathological blood vessels.
Mast cell stabilization. Mast cells are key inflammatory mediators in melasma, releasing histamine, tryptase, and VEGF. TXA has been shown to reduce mast cell degranulation, decreasing the release of these pro-inflammatory and pro-angiogenic mediators. This is particularly relevant because mast cell density is significantly elevated in melasma lesions.
Prostaglandin pathway suppression. TXA inhibits the release of arachidonic acid from keratinocyte cell membranes, reducing the production of prostaglandin E2 (PGE2). PGE2 directly stimulates melanocyte dendricity (the extension of melanocyte processes that transfer melanin to surrounding keratinocytes) and melanin synthesis.
Tyrosinase activity reduction. Through the reduction of UV-induced plasmin activity, TXA indirectly decreases the expression and activity of tyrosinase — the rate-limiting enzyme in melanin synthesis. This provides an anti-melanogenic effect that complements the anti-vascular and anti-inflammatory actions.
The convergence of these mechanisms explains why TXA is effective for melasma while being fundamentally different from traditional depigmenting agents. Rather than blocking a single step in melanin production, TXA addresses the upstream vascular and inflammatory environment that drives the entire melasma process.
Safety Profile and Practical Considerations
Intradermal TXA injection has an excellent safety profile, particularly when compared to oral administration. Because the total dose delivered per session is small (typically 50 to 100 mg total, distributed across the treatment area), systemic absorption is minimal and the theoretical risk of thromboembolic complications is negligible.
Local side effects are mild and transient. Patients may experience temporary erythema (redness) at injection sites, which typically resolves within 2 to 4 hours. Pinpoint injection marks may be visible for 24 to 48 hours. Minor bruising can occur but is infrequent with proper technique and fine-gauge needles.
The Melasma Injection Treatment protocol spaces sessions 3 to 4 weeks apart, allowing adequate time for the biological response to each treatment to develop. This interval also means that there is no cumulative systemic TXA exposure, unlike daily oral dosing which maintains continuous plasma levels.
Contraindications for intradermal TXA are few but important. Patients with a history of thromboembolic disease, active deep vein thrombosis, or known hypercoagulable states should not receive TXA by any route without careful medical evaluation. Pregnancy is a contraindication. Concurrent use of oral contraceptives warrants discussion due to the additive theoretical thromboembolic risk, though the localized delivery method significantly mitigates this concern.
For patients who have been taking oral TXA for melasma, the transition to intradermal injection often allows discontinuation of the oral medication while achieving equal or superior results — eliminating the systemic exposure and the daily compliance requirement simultaneously.
Frequently Asked Questions
Q1: Why not just take oral tranexamic acid pills for melasma?
Oral TXA distributes throughout the entire body, meaning only a tiny fraction reaches the melasma lesion in the dermis. Intradermal injection delivers the full therapeutic concentration directly to the target site. This achieves higher local efficacy with lower total drug exposure and eliminates the systemic side effect risk associated with daily oral dosing.
Q2: Is intradermal TXA injection painful?
The treatment uses very fine needles (30G or 32G), and most patients describe the sensation as mild pinpricks. Topical numbing cream can be applied before the procedure for patients with lower pain thresholds. The entire treatment session typically takes 15 to 30 minutes, and any discomfort resolves immediately after the procedure.
Q3: Can intradermal TXA cause blood clots?
The total dose of TXA used in intradermal injection is extremely small compared to oral dosing — typically 50 to 100 mg per session versus 500 mg per day orally. This minimal dose, combined with local rather than systemic delivery, makes the thromboembolic risk negligible. However, patients with a history of blood clots should disclose this during consultation.
Q4: How does TXA injection compare to laser treatment for melasma?
TXA injection targets the vascular and inflammatory root causes of melasma without generating thermal injury. Lasers target melanin with heat, which can trigger rebound inflammation and worsened pigmentation. For most melasma patients, TXA injection provides more sustainable results with significantly less risk of post-inflammatory hyperpigmentation.
Q5: Can I use topical TXA products alongside the injection treatment?
Topical TXA products can serve as a gentle maintenance tool between injection sessions. However, their limited penetration means they cannot substitute for intradermal delivery. Your physician will advise on incorporating topical products into your between-session skincare routine.
Q6: How soon will I see results from intradermal TXA treatment?
The anti-inflammatory and anti-vascular effects begin immediately, but visible pigment reduction typically becomes apparent after 2 to 3 sessions (approximately 6 to 9 weeks). The vascular component — redness within the melasma — often improves first, followed by gradual lightening of the pigmented component as the inflammatory drive is suppressed and natural pigment turnover proceeds.
About the Author
Dr. Ta-Ju Liu is the founder of Liusmed Clinic and a specialist in regenerative medicine and minimal incision surgery. His approach to melasma treatment integrates pharmacological precision with manual delivery techniques, addressing the vascular and inflammatory foundations of the condition rather than managing surface pigmentation. Liusmed Clinic is dedicated to evidence-based, minimally invasive treatment strategies that produce lasting results.
Disclaimer
This article is provided for educational and informational purposes only. It does not constitute medical advice, diagnosis, or treatment. Individual results vary, and all medical procedures carry inherent risks. Patients with a history of thromboembolic disease, pregnancy, or known hypercoagulable conditions should consult their physician before considering any TXA-based treatment. The information presented reflects clinical experience and published research as of the date of publication.
Specialties
Credentials
- Kaohsiung Medical University, School of Medicine
- Attending Physician, Dermatology, Kaohsiung Chang Gung Memorial Hospital
- Attending Physician, Aesthetic Center, Kaohsiung Chang Gung Memorial Hospital
- Visiting Physician, Dermatology, Xiamen Chang Gung Hospital
- Visiting Physician, Aesthetic Center, Xiamen Chang Gung Hospital
"For every surgery, I strive to achieve the best outcome through the smallest incision and finest technique. Minimally invasive surgery is not just a technique — it's a commitment of respect to every patient."
Recovery from filler complications needs peer support too
Want to learn more?
Schedule a consultation for professional evaluation and advice

