Can You Do Laser or RF Treatments Over Fillers? Melting and Migration Risks
"I Just Had a Radiofrequency Treatment—Why Is My Face Distorted?"
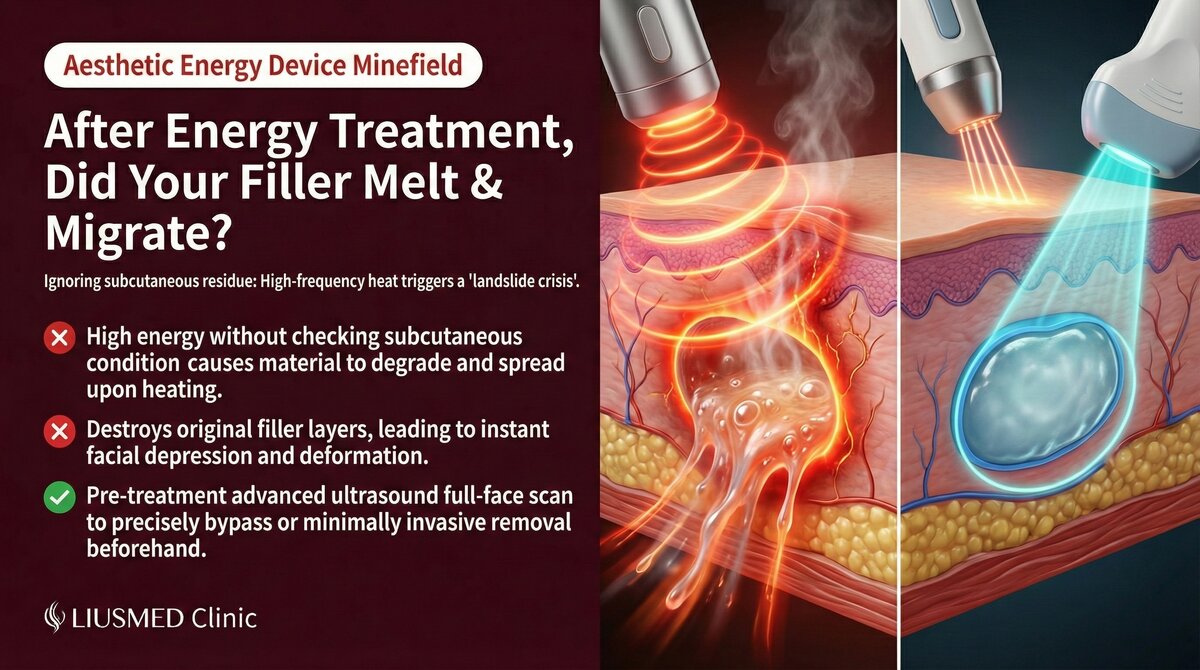
This scenario is becoming increasingly common: a patient had filler injected years ago in a certain area, then visits another clinic for a laser or radiofrequency (RF) treatment. Afterward, the previously injected site shows abnormalities—the filler appears deformed, displaced, or new swelling and lumps have appeared.
The root cause is that energy-based devices (lasers, radiofrequency, ultrasound lifting) generate thermal energy that can directly affect fillers within the body. Many practitioners do not confirm whether patients have fillers before treatment, nor do they understand how different fillers respond to thermal energy.
How Energy Devices Affect Fillers
The Basics of Thermal Effects
Lasers, radiofrequency (RF), and ultrasound lifting devices (such as Ultherapy) share a common mechanism: delivering energy to skin and subcutaneous tissue to produce thermal effects that stimulate collagen remodeling.
- Radiofrequency (RF): Heats the dermal layer to 60-70°C
- Ultrasound lifting: Focused heating to 60-70°C at deeper layers
- Lasers: Heating range from epidermis to dermis depending on wavelength
These temperatures produce controlled effects on normal tissue, but fillers have different physical properties than human tissue and respond differently to thermal energy.
How Different Fillers Respond to Heat
| Filler Type | Heat Sensitivity | Possible Thermal Damage | Risk Level |
|---|---|---|---|
| Hyaluronic acid (HA) | High | Accelerated degradation, liquefaction, displacement | Moderate-high |
| Calcium hydroxylapatite (Radiesse) | Moderate | Carrier gel liquefaction, microsphere aggregation | Moderate |
| Ellanse (PCL) | Moderate-high | Carrier degradation, altered microsphere behavior | Moderate-high |
| Silicone | Low | Chemically stable but surrounding tissue response changes | Moderate |
| PMMA | Low | Microspheres stable but carrier and surrounding tissue affected | Moderate |
| Fat grafting | High | Partial necrosis, liquefaction | High |
Key Insight: Even when a filler's chemical structure is unaffected by heat, thermal reactions in surrounding tissue can cause indirect problems. Heat-induced local vasodilation, tissue edema, and inflammatory responses can all alter filler position and condition.
Specific Clinical Risks
Risk 1: Filler Softening and Displacement
Thermal energy accelerates cross-link breakdown in certain fillers (especially HA), causing softening. Softened filler is more susceptible to displacement under changes in surrounding tissue pressure.
For more on filler displacement mechanisms: Why Fillers Migrate.
Risk 2: Uneven Degradation Causing Appearance Irregularities
Thermal energy distribution within tissue is not perfectly uniform. Filler may be partially degraded in some areas while remaining intact in others, turning previously smooth augmentation into an uneven surface.
Risk 3: Triggering Encapsulation Reactions
Applying thermal stimulation to areas containing existing fillers may activate local immune responses, promoting fibrosis and capsule formation. This can create new problems around previously stable filler.
For detailed analysis of encapsulation: Encapsulation: Why Dissolvers Fail.
Risk 4: Increased Infection Risk
If low-grade biofilm infection already exists around filler (the patient may be unaware), thermal stimulation may disrupt the existing equilibrium, causing latent infection to acutely flare.
Risk 5: Deep Tissue Injury
Focused ultrasound devices can reach the SMAS layer (4.5mm). If filler exists at that depth, focused high temperatures can cause severe localized tissue damage.
| Energy Device | Primary Risk | Depth of Effect | Fillers of Greatest Concern |
|---|---|---|---|
| RF (Thermage, etc.) | Softening, displacement | Dermis to superficial subcutaneous | HA, fat grafting |
| Ultrasound lifting (Ultherapy, etc.) | Deep tissue injury | Dermis to SMAS layer | All deep-layer fillers |
| Fractional laser | Surface degradation | Epidermis to superficial dermis | Superficial HA |
| Intense pulsed light (IPL) | Local heating | Epidermis to superficial dermis | Lower risk |
| Picosecond laser | Minimal thermal effect | Epidermis to dermis | Lower risk but not zero |
Why Many People Only Learn About These Risks After Something Goes Wrong
Incomplete Medical History
Many pre-treatment questionnaires for energy devices do not ask about filler injection history, or ask only about "recent injections" while ignoring treatments from years ago.
Patient Memory Gaps
Many patients do not remember what material was injected years ago or have even forgotten they received injections at all. This is particularly common after "medical tourism."
Practitioner Knowledge Gaps
Some practitioners are unaware of the potential effects of energy devices on fillers, assuming filler injections and light/energy treatments do not interact.
Key Insight: "Not knowing there is filler in the body" does not mean "there is no filler in the body." Many fillers—especially Radiesse, Ellanse, and silicone—persist far longer than patients expect. Material injected years or even a decade ago may still be in place.
What Should Be Done Before Treatment
Comprehensive Treatment History Review
Before any energy device treatment, detailed inquiry should cover:
- All previously injected sites and timing
- Material injected (if known)
- Dosage and depth of injection (if known)
- Whether any existing lumps or abnormalities are present
The Necessity of Ultrasound Examination
For patients with injection history but uncertain about residual status, ultrasound can:
- Confirm whether filler material remains
- Identify filler type and location
- Assess whether encapsulation or other abnormalities exist
- Provide a safety reference for subsequent energy treatments
For more on the ultrasound evaluation process: Filler Repair Evaluation Process.
What to Do If Energy Treatment Has Already Been Performed Over Fillers
If energy device treatment has already been performed on an area containing filler and problems have emerged:
- Stop further treatments—do not continue using energy devices in that area
- Document symptom changes—photograph appearance changes, swelling, texture alterations
- Seek professional evaluation—ultrasound examination to confirm filler status
- Determine management based on findings—may include observation, medication, or filler removal
Schedule a consultation for professional evaluation and the most appropriate management plan.
Conclusion
Energy devices and fillers are each effective cosmetic tools in their own right, but their interaction is a seriously overlooked safety issue. In an era where nearly everyone has some history of injections or energy treatments, "confirm before treating" is not excessive caution—it is basic respect for patient safety.
Key Insight: The safest treatment is not necessarily the newest device or highest energy setting—it is treatment built on complete information. Knowing what is inside the patient's body, where it is, and what condition it is in—this is the starting point for all subsequent treatment decisions. "See before you treat" is especially critical when navigating the interaction between energy devices and fillers.
Related Services
Specialties
Credentials
- Kaohsiung Medical University, School of Medicine
- Attending Physician, Dermatology, Kaohsiung Chang Gung Memorial Hospital
- Attending Physician, Aesthetic Center, Kaohsiung Chang Gung Memorial Hospital
- Visiting Physician, Dermatology, Xiamen Chang Gung Hospital
- Visiting Physician, Aesthetic Center, Xiamen Chang Gung Hospital
"For every surgery, I strive to achieve the best outcome through the smallest incision and finest technique. Minimally invasive surgery is not just a technique — it's a commitment of respect to every patient."
Recovery from filler complications needs peer support too
Want to learn more?
Schedule a consultation for professional evaluation and advice

